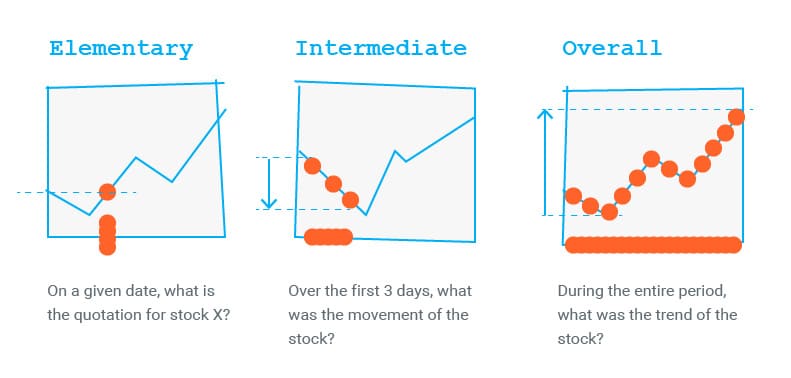
![Consider the following mechani[{Image src='img137723731702631852348.jpg' alt='' caption=''}]sm. Determine the rate law for the overall reaction (where the overall rate constant is represented as k). | Homework.Study.com Consider the following mechani[{Image src='img137723731702631852348.jpg' alt='' caption=''}]sm. Determine the rate law for the overall reaction (where the overall rate constant is represented as k). | Homework.Study.com](https://homework.study.com/cimages/multimages/16/img137723731702631852348.jpg)
Consider the following mechani[{Image src='img137723731702631852348.jpg' alt='' caption=''}]sm. Determine the rate law for the overall reaction (where the overall rate constant is represented as k). | Homework.Study.com

Determine the rate law for the overall reaction (where the overall rate constant is represented as k)? | Socratic

SOLVED: Consider the mechanism Step 1: A = B+C Step 2: C+D-E Overall: A +D - B+E equilibrium slow Determine the rate law for the overall reaction, where the overall rate constant

what effect does a decrease in temperature have on the overall rate of a chemical reaction?? A - Brainly.com

The process starts with problem identification on an overall level to... | Download Scientific Diagram

Consider the following mechanism:Step 1: 2A ----> B slow.Step 2: B + C ----> D fast - Home Work Help - Learn CBSE Forum
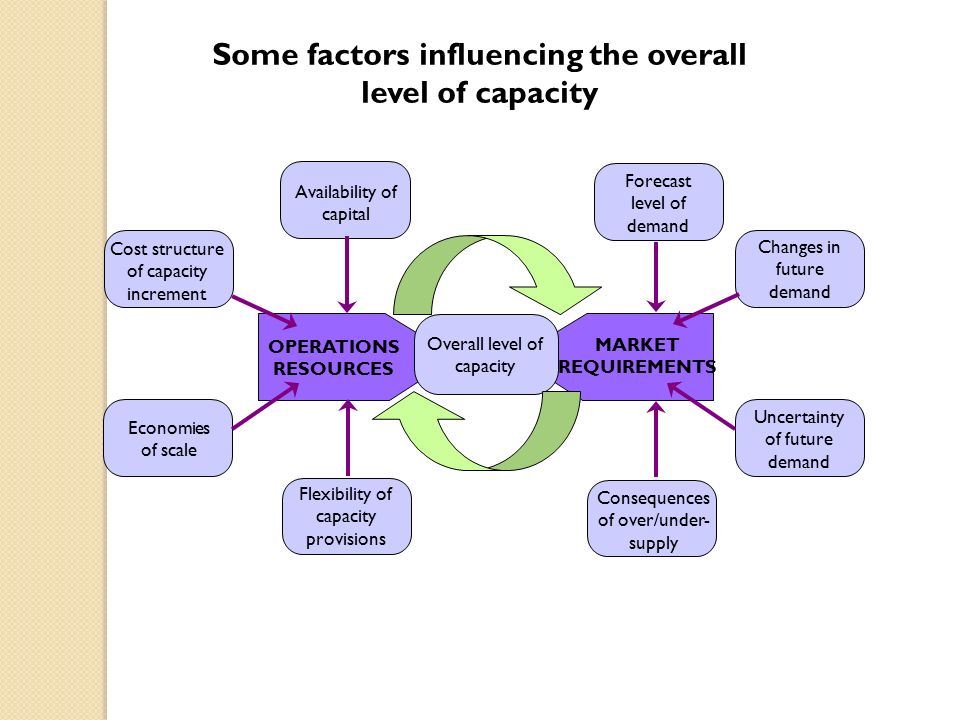
Capacity strategy. Some factors influencing the overall level of capacity Forecast level of demand Consequences of over/under- supply Availability of. - ppt download

Determine the rate law for the overall reaction (where the overall rate constant is represented as k) - Home Work Help - Learn CBSE Forum

Steelers, Canada Sticking To The Plan In QB Competition, Says 'We've Seen' In-Stadium Results In Practice - Steelers Depot

/cloudfront-us-east-1.images.arcpublishing.com/gray/CNUDMZRM55GCNIR32B2EETXSNI.JPG)