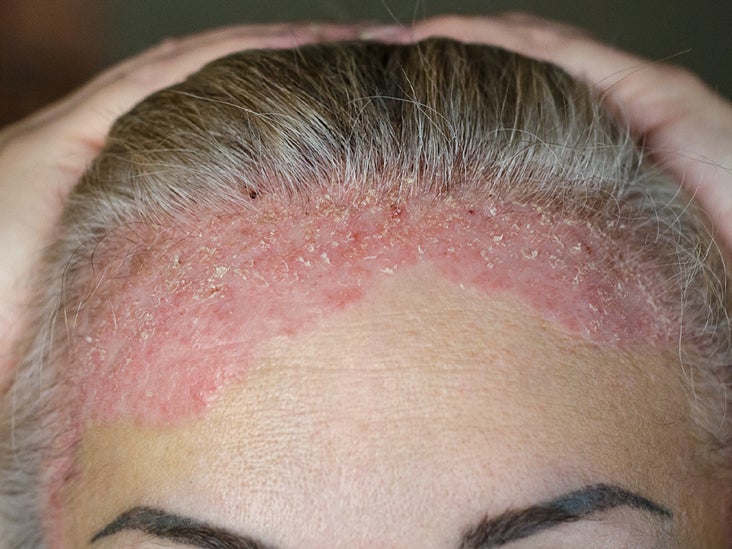
Improvement in Psoriasis Symptoms and Physical Functioning with Secukinumab Compared with Placebo and Etanercept in Subjects with Moderate-to-Severe Plaque Psoriasis and Psoriatic Arthritis: Results of a Subanalysis from the Phase 3 Fixture
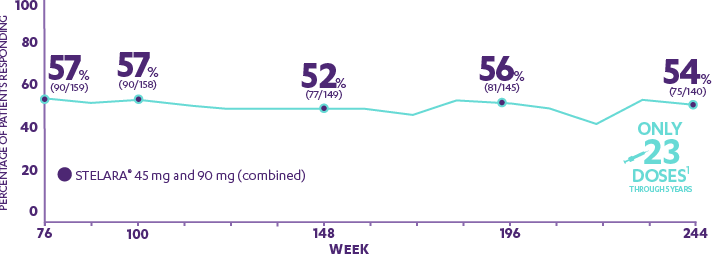
New Novartis data shows Cosentyx™ is significantly superior to Stelara® and clears skin (PASI 90) in nearly 80% of psoriasis patients | Novartis

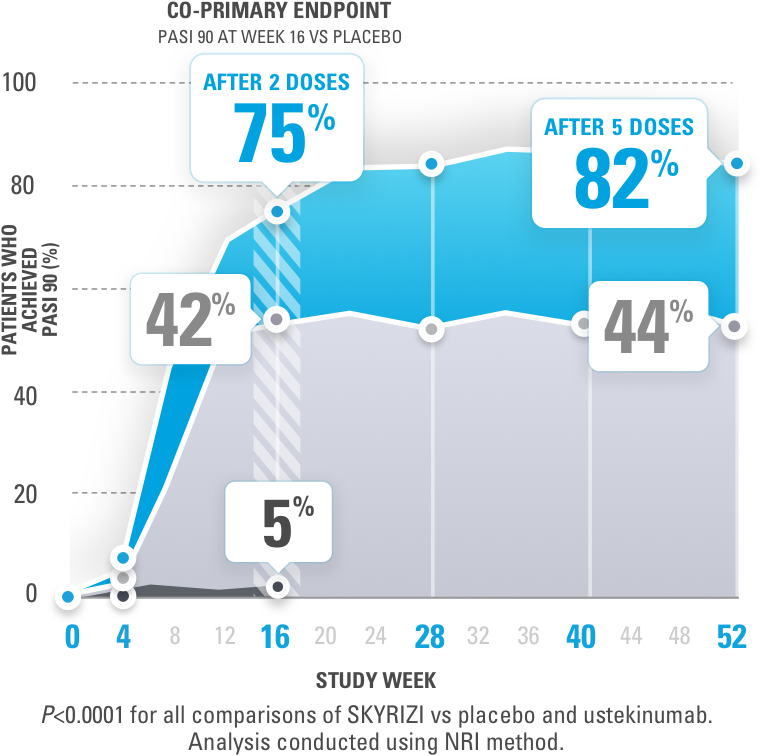
Exposure–Response Relationships for Efficacy and Safety of Risankizumab in Phase II and III Trials in Psoriasis Patients - Khatri - 2020 - Clinical Pharmacology & Therapeutics - Wiley Online Library

Figure 2 | Exposure–Response Relationships for the Efficacy and Safety of Risankizumab in Japanese Subjects with Psoriasis | SpringerLink

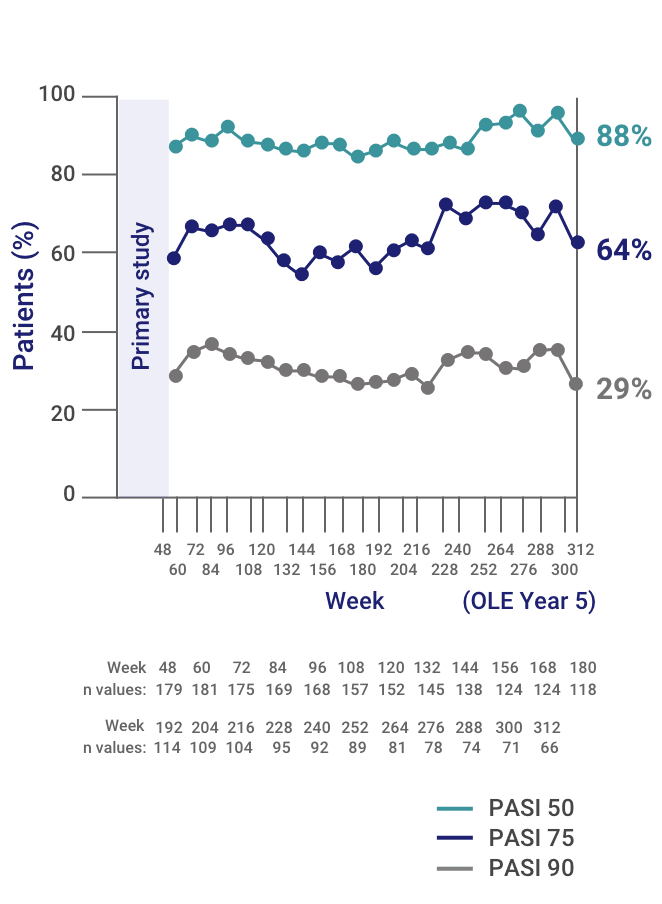
Ixekizumab sustains high level of efficacy and favourable safety profile over 4 years in patients with moderate psoriasis: results from UNCOVER‐3 study - Lebwohl - 2020 - Journal of the European Academy

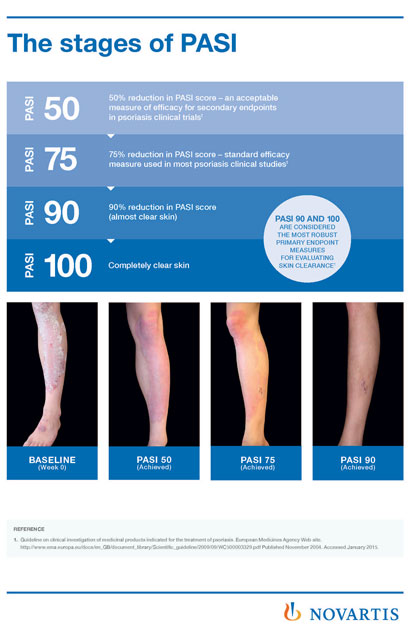
Absolute Versus Relative Psoriasis Area and Severity Index in Clinical Practice | Actas Dermo-Sifiliográficas
Real-world efficacy of biological agents in moderate-to-severe plaque psoriasis: An analysis of 75 patients in Taiwan | PLOS ONE
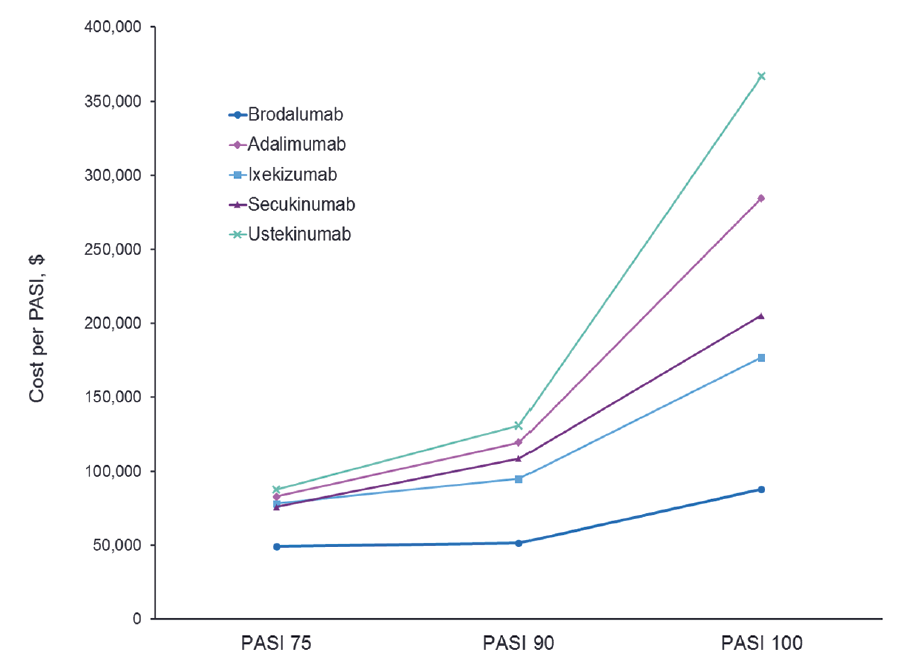
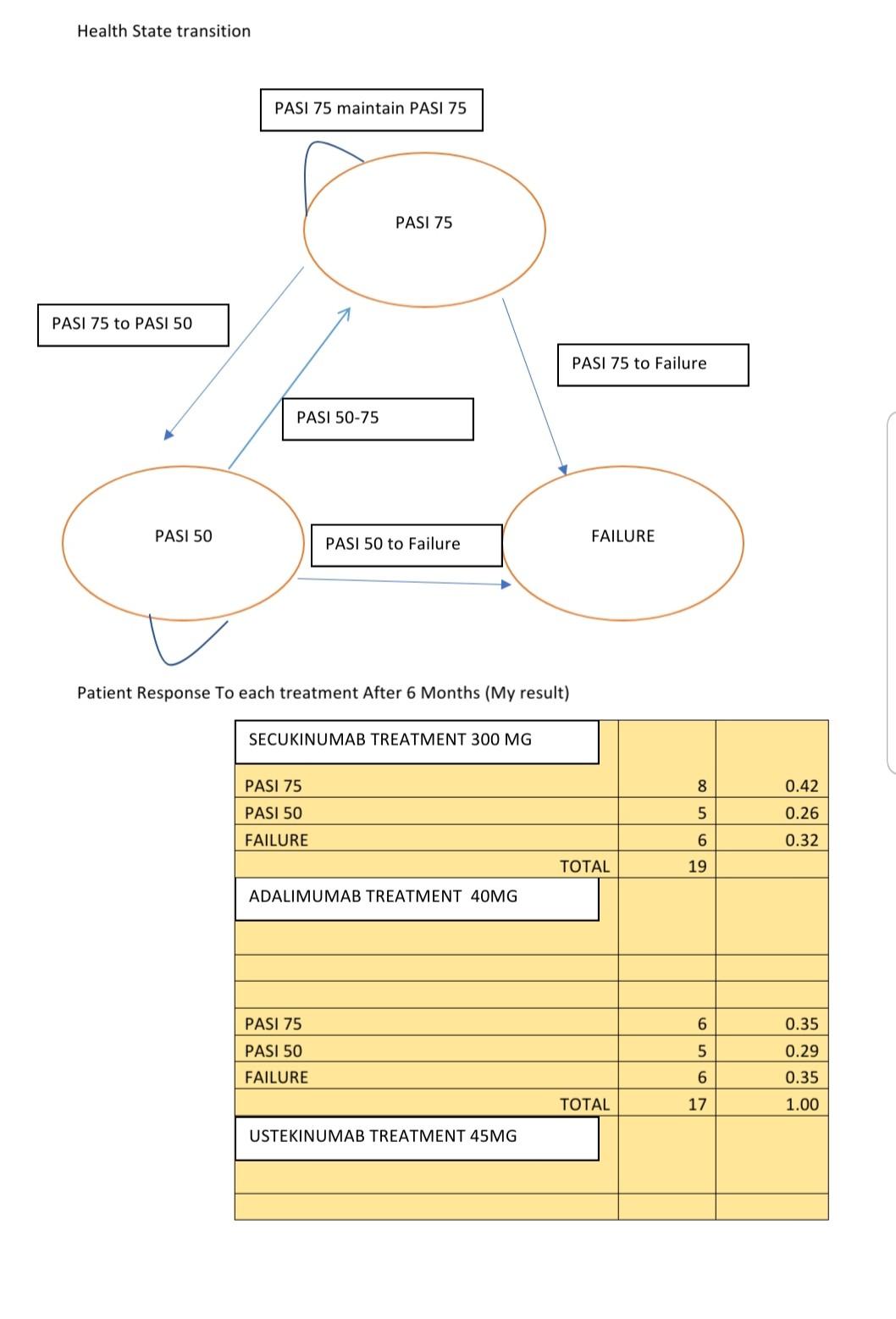
What Does Efficacy Cost? Evidence for Relative Cost-Effectiveness of Biologic Therapies for Psoriasis - Practical Dermatology
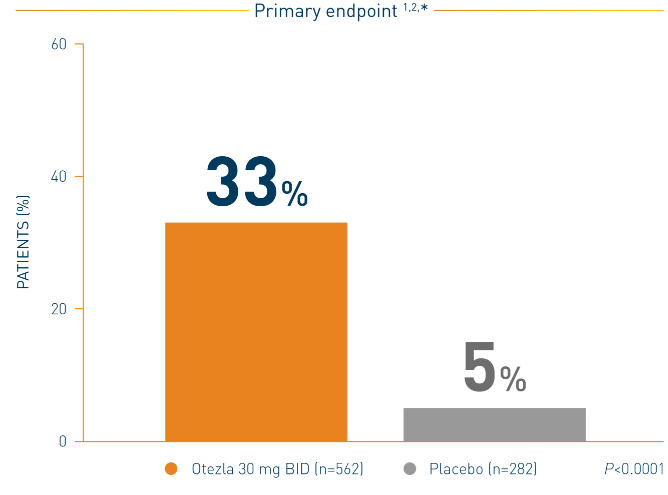
PASI-75 Response with Otezla in the Treatment of Moderate to Severe Plaque Psoriasis — Efficacy | Otezla® (apremilast) Healthcare Professional Site

JCM | Free Full-Text | Use of Guselkumab for the Treatment of Moderate-to-Severe Plaque Psoriasis: A 1 Year Real-Life Study | HTML

Ixekizumab, an interleukin-17A specific monoclonal antibody, for the treatment of biologic-naive patients with active psoriatic arthritis: results from the 24-week randomised, double-blind, placebo-controlled and active (adalimumab)-controlled period ...